Project EARTH (Environmentally Applied Research Toward Hydrofluorocarbons) is focused on identifying sustainable processes for the selective separation of hydrofluorocarbon (HFC) refrigerant mixtures. Currently, there are millions of kilograms of HFC refrigerant mixtures in use worldwide. These refrigerants have zero ozone depletion potential but many HFCs have high global warming potential (GWP). This has led to recent legislation restricting the use of some HFC refrigerants in particular applications and the eventual phase-out of HFC refrigerants. Consequently, phase-out means that industries across the world will need to dispose of their current HFC refrigerants. Rather than incinerate/vent these high GWP refrigerants to the atmosphere, a process which would both harm the environment and waste millions of kilograms of refrigerants, these refrigerant mixtures could be recycled. To accomplish this, the HFC refrigerant mixtures would first need to be separated into their constituent refrigerants which can be especially difficult because many of the HFC mixtures form azeotropes. Project EARTH members are working to address this difficult separation challenge using the following three separation techniques:
Ionic Liquids: The ionic liquid (IL) portion of Project EARTH is a collaboration project for the measurement and simulation of HFC mixture properties in ILs between the University of Kansas and the University of Notre Dame. The University of Kansas is focused on the material discovery and property measurements of HFCs and ILs and the University of Notre Dame is focused on Monte Carlo simulations and process modeling/optimization of ILs and HFC/IL mixtures. ILs are of particular interest in this project because of their unique properties including: no measurable vapor pressure, dissolution of many organic and inorganic compounds, variable solubility of gases and liquids, and high thermal, chemical, and electrochemical stability. These properties have allowed ILs to be used in many different applications including separation and purification processes. Our goal is to determine the most efficient ILs for the HFC separations.
Membranes: Membranes provide a unique opportunity for the separation of HFC refrigerant components, as membrane technology has the potential of having smaller energy and capital requirements than traditional separation techniques. Membranes can be described as a barrier that selectively allows the passage of some species while preventing the passage of others. While semipermeable polymers are the most common membranes, different components and compositions can be incorporated to influence membrane selectivity and permeability, thus making it possible to design membranes capable of difficult HFC separations. While much research has been devoted to the separation of refrigerants from air streams, there has been little exploration of membrane technology for the separation of refrigerant mixtures. In the membrane division of Project EARTH, various types of membranes and membrane compositions will be explored for separating HFCs.
Porous Media: The term porous media applies to various different nano to micro-sized materials such as zeolites, activated carbons (ACs), and metal organic frameworks (MOFs). Dr. David R. Corbin (DuPont Senior Research Fellow, retired) with over forty years of zeolite experience is working in our group as a Senior Scientist assisting with Project EARTH. Zeolites are hydrous, aluminosilicate, porous crystals which are used extensively in industry for both catalysis and molecular separations. Due to their interconnected, uniform pores, zeolites can be exploited for both their molecular sieve capabilities and chemical properties. Zeolites are therefore a good candidate for performing the challenging separations which are required for HFC refrigerant mixtures.
Project EARTH team members:
University of Kansas
Kalin R. Baca, Abby N. Harders, Andrew D. Yancey, Greta M. Olsen, Dr. Berlyn R. Mellein, Dr. David R. Corbin, and Prof. Mark B. Shiflett
Rutgers University
Prof. Ed Castner
University of Notre Dame
Bridgette Befort, Prof. Alex Dowling, and Prof. Ed Maginn
Texas A&M University
Randinu Pulukkody, Mohammed Sadaf Monjur, Ashfaq Iftakher, Prof. Faruque Hasan, and Prof. Emily Pentzer
Chemours
Dr. Luke Simoni
Hiden
Dr. Mike Benham
Iolitec
Dr. Thomas Schubert
Critical Metals Recovery Using Oxalate Chemistry
Sustainable, energy-efficient technlology for critical metals recovery is urgently needed across industries dealing with mixed metal sources such as lithium-ion batteries (LIBs), ore processing, catalyst manufacturing, and rare earth element (REE) sourcing. Oxalate, which can be derived from organic sources, has minimal to no environmental effects, is generally safer to work with than typical inorganic acids, and may offer the opportunity for developing a more energy-efficient process. The Shiflett group has identified oxalate as a sustainable reagent that can provide both the desired selectivity and efficient leaching capabilities for a variety of mixed metals under mild reaction conditions. The variation in the solubility of different metal oxalate species can be utilized effectively for the separation of metals.
Currently, the Shiflett group is working on the development of an oxalic acid based closed-loop process for the recycling of cathodes from lithium-ion batteries. In this work, lithium and cobalt, two of the critical metals are recovered and separated from the spent lithium cobalt oxide (most common cathode of lithium-ion batteries). In traditional approaches using sulfuric acid and nitric acid with a reducing agent like H2O2, both lithium and cobalt completely dissolve into the solution. The metal separation from leachate requires another precipitation step which adds to the overall cost of the process. Also, the emission of harmful pollutants such as SOX and NOX pose a significant environmental challenge. All of these issues can be eliminated using oxalic acid and its derivatives for this process. In the oxalic acid process, cobalt directly precipitates out as cobalt oxalate which can be converted to cobalt oxide by appropriate thermal treatment, whereas lithium present in the solution can be precipitated out in the form of carbonate or phosphate. Remaining oxalate in the filtrate after lithium precipitation can be recycled and used for extraction again which provides a closed-loop process. The possibility of reusing the filtrate provides an economic advantage to the oxalate chemistry. This chemistry has tremendous potential for application to other metal recovery processes from various mixed metal oxide sources like ores, spent catalysts and other types of spent LiBs cathodes.

Project SAVE: Silica Adsorbed Vaccine Encapsulation

The refrigeration of vaccine is critical to maintaining their efficacy. Vaccines are mainly composed of proteins, most of which are very sensitive to temperature changes. The cold chain cycle assures that vaccines are stored in controlled environments, between 2 to 8°C, to avoid conformational changes in protein structures. Keeping a constant environment that is optimal to the vaccine reduces changes to the protein’s secondary and tertiary structures while maintaining the vaccine’s potency. Any deviation from this range can result in significant waste and lead vaccination programs to a deficit. This is a common concern especially in developing countries where access to the necessary resources to store vaccines and a constant source of electricity are limited.


In the Shiflett group, we are currently trying to eliminate a vaccine’s dependency on refrigeration to reduce waste, cost, and increase the access of vaccines to those who need them. To accomplish this, we are currently working with Dr. David Corbin (Senior Scientist, CEBC) and Dr. William Picking (Distinguished Professor, KU) on selecting and developing vaccine specific inorganic vehicles that will act as thermal stabilizers and enhancers due to their adjuvant properties. Dr. William Picking, an expert in vaccines and pathogenic microbiology, completes our team by integrating his knowledge of pathogens, as we work to characterize the interactions between the inorganic vehicles and the Invasion Plasmid Antigen D. Together we are looking to apply our knowledge and resources to thermally stabilize vaccines in tailored inorganic materials.
Project SAVE team members: Nicole Montoya, Rhianna Roth, Elizabeth Funk, Dr. Philip Gao, Dr. David Corbin, Dr. Mark Shiflett.
Explosion Suppression of Metal Dust Deflagrations
Combustible dust explosions continue to present a significant threat toward an extensive range of industries processing, storing, or pneumatically conveying metal dust hazards. Upon ignition within a contained enclosure volume and propagation of flame toward interconnected vessels, metal dust deflagrations demonstrate an extremely reactive explosion risk relative to organic dusts due to well-documented amplified heat of combustion, burning temperature, flame speed, rate of pressure rise, maximum explosion overpressure, and ignition sensitivity. Addition of non-combustible inert material to combustible dust mixtures, either through pre-mixing or high-rate injection as the incipient flame front begins to develop, is common practice for preventative inhibition or explosion protection via active suppression, respectively. However, inhibition efficiency of suppressant agents utilized for active mitigation is shown to be reliant on comparative explosibility, discrete burning mechanism, and combustion temperature range, and thus may be increasingly variable depending on the fuel in question. For this reason, mitigation of metal powder deflagrations at moderate total suppressed pressures (relative to the overall strength of the enclosure) and at low agent concentrations has remained challenging. This report reviews applicable thermal analytical techniques and large-scale suppression testing in Fike Corporation’s 1 m3 sphere combustion chamber to evaluate the efficacy of multiple suppressant agents for the mitigation of contained iron and aluminum powder deflagrations.
Through recent years, investigations have thoroughly observed the influence of particle size, polydispersity, and chemical composition on dust explosion sensitivity and severity. However, studies characterizing the effect of particle shape (or morphology) on metal dust explosibility are limited and merit further consideration in order to better define the hazard and understand unique allowances for implementation of protection. In this work, high-purity aluminum dust samples of three unique particle morphologies were examined (spherical, irregular, and flake). Investigations performed in a Kühner MIKE3 minimum ignition energy apparatus and a Siwek 20 L sphere combustion chamber resulted in the direct characterization of explosion sensitivity and severity, respectively, as a function of suspended fuel concentration and variable particle morphology. Such fuel reactivity shall be modeled as a means of predicting hazardous potential for distinct metal dust processing methods. Applying the shrinking particle theory with reaction and species diffusion limitations, previously reported pressure evolution outcomes were verified through development and implementation of closed-vessel numerical modeling reliant on fundamental mass and thermal balance equations.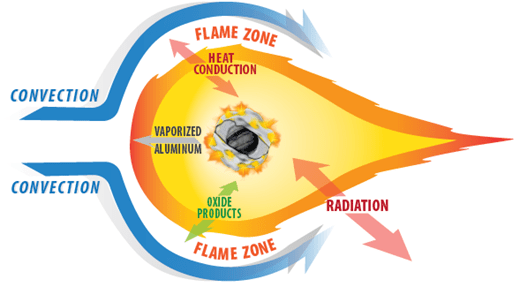
Removal of PFOA from Drinking Water Using Microporous Zeolites
Local, state, and federal governments are responding to perfluoroalkyl substances (PFAS) that are being detected in drinking water. These persistent materials are  bioaccumulative and maybe linked to human and animal health problems. The State of New Jersey recently set the first drinking water standard in the U.S. for perfluorononanoic acid (PFNA) at 13 parts per trillion (ppt) which is only one of many known PFAS. Other common PFAS include perfluorooctanoic acid (PFOA) and perfluorooctanoic sulfate (PFOS). PFAO has been used in the production of a variety of materials such as nonstick Teflon™ cookware, firefighting foam and water repellant coatings. Recently, high levels of PFOA and PFOS were detected in the drinking water in Parchment, Michigan. The state’s Department of Environmental Quality (DEQ) results showed water with 1,410 ppt (740 ppt PFOS and 670 ppt PFOA). Michigan in January adopted 70 ppt as a legally enforceable level for PFOS or PFOA in drinking water. Today, almost every week a new story about drinking water contaminated with PFAS is appearing in the news. The U.S. Environmental Protection Agency (EPA) is recommending that drinking water should contain less than 70 ng/L (70 ppt) of PFOA. The Shiflett group is studying a variety of materials for removing PFOA, PFOS, and other PFAS compounds from drinking water. They have recently discovered a family of zeolites that can remove PFOA from aqueous solutions that may be more cost effective than activated carbons. Along with the removal of PFOA from drinking water, the team is working with the Department of Chemistry to develop an ultra-performance liquid chromatography (UPLC) triple quad mass spectrometer (MS) technique (UPLC-MS) for detection of PFOA and other PFAS materials below 10 ppt.
bioaccumulative and maybe linked to human and animal health problems. The State of New Jersey recently set the first drinking water standard in the U.S. for perfluorononanoic acid (PFNA) at 13 parts per trillion (ppt) which is only one of many known PFAS. Other common PFAS include perfluorooctanoic acid (PFOA) and perfluorooctanoic sulfate (PFOS). PFAO has been used in the production of a variety of materials such as nonstick Teflon™ cookware, firefighting foam and water repellant coatings. Recently, high levels of PFOA and PFOS were detected in the drinking water in Parchment, Michigan. The state’s Department of Environmental Quality (DEQ) results showed water with 1,410 ppt (740 ppt PFOS and 670 ppt PFOA). Michigan in January adopted 70 ppt as a legally enforceable level for PFOS or PFOA in drinking water. Today, almost every week a new story about drinking water contaminated with PFAS is appearing in the news. The U.S. Environmental Protection Agency (EPA) is recommending that drinking water should contain less than 70 ng/L (70 ppt) of PFOA. The Shiflett group is studying a variety of materials for removing PFOA, PFOS, and other PFAS compounds from drinking water. They have recently discovered a family of zeolites that can remove PFOA from aqueous solutions that may be more cost effective than activated carbons. Along with the removal of PFOA from drinking water, the team is working with the Department of Chemistry to develop an ultra-performance liquid chromatography (UPLC) triple quad mass spectrometer (MS) technique (UPLC-MS) for detection of PFOA and other PFAS materials below 10 ppt.
Designing Molecular Gate Adsorbents for Natural Gas Purification using PSA
The PSA system is used to study the separation of contaminants such as N2, CO2, CO, and H2S from natural gas (NG). Shiflett lab group is currently working with Dr. David Corbin (Senior Scientist, CEBC) to develop new molecular sieves for the kinetic separation of N2, CO2, CO, and H2S from NG.

To remove contaminants, the natural gas is fed under pressure through a PSA column containing molecular sieve which can adsorb N2, CO2, CO, and H2S. After the sieve is saturated with N2, CO2, CO, or H2S, the flow is redirected to the next column and the first column is depressurized to desorb the N2, CO2, CO, and H2S and regenerate the sieve. In some cases the desorption is slower than the adsorption step, so multiple columns are needed to allow time for complete column regeneration. Development of a molecular sieve to remove N2, CO2, CO, and H2S from CH4 based on size difference is challenging because they only differ in size by 0.2 Angstroms (N2, CO2, CO, and H2S are about 3.4 to 3.6 Angstroms vs. CH4 is 3.8 Angstroms). However, if a molecular sieve can be developed for this application, it will significantly reduce the cost of separating inert gases such as N2, CO2, CO, and H2S and possibly He from NG. Current technologies, such as cryogenic plants for N2 removal, are complex and have high capital and operating costs compared to using a PSA system. The PSA technology is also field deployable, modular and can be scaled out to meet the capacity of the gas field.
Sulfite Removal from Wine

Within the winemaking industry, sulfites are used as a chemical additive to prevent wines from spoiling or browning as they age, and to ensure that wines are free of unwanted bacteria at the time of opening. While there are obvious advantages to using sulfites in wine, a small percentage of the population experiences severe adverse health effects as a result of a sulfite sensitivity. These people may experience asthma attacks, rashes, and headaches as a results of the presence of sulfite in wine. Our research group is designing a device to go on the end of a wine bottle to remove sulfites from wine at the time of pouring. Students are working to design and print a 3D prototype, while also investigating materials such as zeolites, ion exchange resins, and clays to discover which materials may selectively remove sulfite without changing the taste of wine. Students are using a combination of qualitative and quantitative techniques to investigate which materials may be more successful at removing sulfites than the few commercial products that already exist on the market. It is the hope of our group to design a cost-effective single use device to allow wine drinkers to enjoy wine without the presence of unnecessary chemical additives.
Ammonia Absorption in Room Temperature Ionic Liquids
Ionic liquids are relatively new generation solvents with low vapor pressure and high thermal stability. The solvency of the ionic liquids can be tuned by the variation in ion types, substitution and composition to provide unique properties to many industrial applications. In our research group, we have over 100 ionic liquids in our library.
Ammonia water vapor absorption system is an ancient heating/cooling system for large industrial applications. The physical properties of ammonia provide efficient, low cost cooling for large systems. The major drawback of the current system is the high cost of separation of water and ammonia in a separation unit. Ionic liquids with low vapor pressure offer an opportunity to overcome this problem and lower the operation costs. Our research group’s goal is to utilize ionic liquids to provide a high efficiency and economic alternative to the current system.
Water Desalination via Gas Hydrate Technology
Global population continues to increase, consequently the demand for freshwater keeps rising. As a response to this pressing need, several desalination plants have developed during the past decades. Most of these utilize reverse osmosis (RO) or multi-stage flash distillation (MSF) as their separation technology. However, pumping large amounts of liquids to elevated pressures and the significant thermal heating requirement makes these technologies expensive. An alternative desalination technology is the use of clathrate hydrates.

A clathrate hydrate is a crystalline cage-like water structure which encloses a guest molecule. The conditions at which clathrate hydrates form are specific to the guest molecule. In past experiments, researchers have investigated several different refrigerants (HCFC-22, HFC-134A, HCFC-141b, etc.) as clathrate formers to separate brine from freshwater and have observed clathrates form at temperatures above the freezing point of water. This is an important benefit because it decreases the energy needed to cool the seawater to reach crystallization conditions. Nonetheless, due to the ozone depletion and global warming potentials related to CFCs, HFCs, and HCFCs, the clathrate formers investigated in the past are no longer environmentally viable.
The purpose of our research is to identify an optimum clathrate former. We will achieve our goal through fundamental studies on phase transition properties, hydrate formation kinetics, and solubility of refrigerants in saline water, while simultaneously sustaining a pragmatic approach to desalinating water by considering heat integration and technology coupling.


You must be logged in to post a comment.